Gut Health and Pain – Part 1: Know Your Gut
Over the past decade there has been a lot of discussion about the importance of our gut. Our gut has been shown to be one of the keys to our overall health and wellbeing.
- It has been called our “second brain” due to the intricate nervous system (the enteric nervous system) it holds within in and how it communicates with our brain (the gut-brain axis).
- It has been shown to play a vital role in the production of serotonin, which has been one of the keys to understanding the role it plays in mood and sleep.
- It has been shown to play a vital role in our stress (hypothalamic pituitary adrenal (HPA) axis) and relaxation (through the vagus nerve) response.
- It is home to 100 trillion microorganisms and their genetic material, known as microbiome, and scientists are only just unlocking the potential of this live bacteria.
- Some of the most interesting areas being explored by scientists include the role our gut plays in immunity, inflammation and pain.
The following series “Gut Health and Pain” will take a look at the intricate world of our gut and unravel the mystery of why there is thousands journal articles being written each year about the health of our gut and the problems an unhealthy gut can cause. Welcome to Part 1 – Know Your Gut.

Part 1 – Know your Gut
To understand the importance of our gut and the role it plays in our health and wellbeing, it is important to get to know our gut – what it is, what it does, why it matters and how it relates to pain. In this article we will discuss:
- What is your gut?
- What is gut microbiome?
- What is the link between our gut microbiome and pain?
- How does the lining of our gut support our immune system?
- What is the link between our gut, immune system and pain?
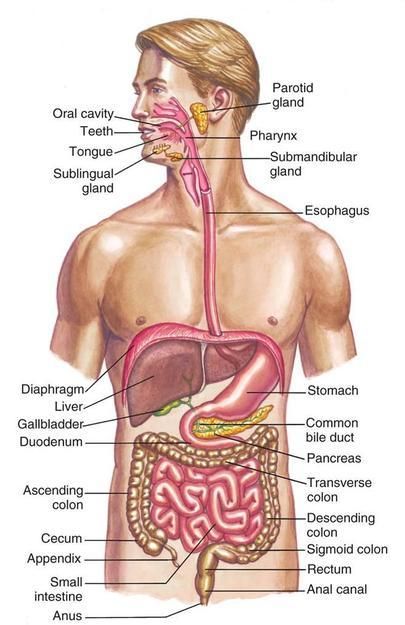 What is your gut?
What is your gut?
Our “gut” is also known as the gastrointestinal system, gastrointestinal tract, digestive system and digestive tract. It is made up of a group of organs which include: our mouth, salivary glands, oesophagus, stomach, panaceas, liver, gallbladder, appendix, small intestine, large intestine, rectum and anus.
Our gut uses the process of digestion to break down the food we eat into the nutrients and energy we need to live and thrive. At each level of our digestive system – from our mouth to large intestine – food is being broken down and converted, so that we can breathe, concentrate, build muscle, repair, grow and survive. The saying “you are what you eat” is true: what we eat and drink becomes the foundation of our overall health and wellbeing, making up the structure, function and integrity of every single cell in our bodies.
The food we eat is used for:
- Brain and memory function
- Eye function
- Digestion and transportation of waste products
- Heart and blood health
- Bone, muscle and ligament repair and strengthening
- Energy for mental and physical activities
- Building and strengthening our immune system
- Repairing cells and tissues
As the below video explains, over 40-50 hours our gut converts the food we eat into the nutrients, vitamins and minerals we need to get through the day. But food conversion is only one piece of the puzzle.
Video courtesy of the TedEd
Our gut works as a guard and barrier: keeping the contents within, from escaping into the bloodstream until they are ready and safe to do so. Our gut also performs many other vital functions including:
- Sustaining and protecting our overall health and wellbeing
- Allowing us to absorb water and nutrients from the food we eat
- Converting food into the building blocks our bodies need to live, function and stay healthy
- Producing and/or hosting many of our neurotransmitters – including serotonin and GABBA
- Supporting our immune system
- Communicating with our brain (the gut-brain axis)
- Playing host to more than 100 million nerve cells (The Enteric Nervous System)
- Modulating stress, mood, pain and our general state of mind
- Feeding and hosting our gut microbiome
What is our gut microbiome?
Our gut microbiome is made up of the 100 trillion microorganisms and their genetic material which live within our gut. These micro-organisms include: fungi and bacteria. Predominantly our microbiome is made up of bacteria and this varies from person to person.
Factors that play a role in the type, variety and quantity of our microbiome include:
 How you were born (caesarean versus vaginal)
How you were born (caesarean versus vaginal)- Your first foods as a baby
- Your age and gender
- Where you live
- Your hygiene
- Your stress level
- Medications you take, particularly antibiotics
- Any other diseases or illness you may have
All of these factors make up our “unique bacterial fingerprint”. This unique bacterial fingerprint serves many vital functions which support our overall health and wellbeing, including:
- Supporting immune function
- Digestion of the food you eat and the nutrients you absorption, in particular iron, zinc and magnesium
- Healthy energy production
- Controlling inflammation
- Improving mood, in particular through production of serotonin
- Vitamin production such as folate, biotin, B12 and K2
- Managing your metabolism and weight
- Improving your sleep
- Managing pain
A healthy gut has a diverse and abundant microbial colony and is known as “Eubiosis“. A low, irregular or poor balance of microbiome – known as “dysbiosis” – has been linked to many disease states including depression, inflammatory bowel disease, fibromyalgia, complex regional pain syndrome, multiple sclerosis, autism spectrum disorder and Alzheimer’s disease. This is an area that researchers are looking into with great interest as it is still difficult to ascertain if the dysbiosis is a cause or result of these diseases.
Video courtesy of the conversation
What is the link between our gut microbiome and pain?
Changes to gut bacteria in people with painful gastrointestinal issues such as inflammatory bowel disease and irritable bowel disease, has long been recognised. However, emerging research looking into specific painful conditions such as fibromyalgia, inflammatory pain, headache, neuropathic pain and even opioid tolerance, has helped reinforce the relationship gut bacteria has with other forms of chronic pain.
Researchers in this area believe that signal-carrying molecules – which are by-products of microbiota (the cells which are made up of your microbiome), metabolites (comes directly from our food), neurotransmitters, and neuromodulators – regulate peripheral and central sensitisation. This has an impact on the development and chronicity of pain. These researchers propose that changes to diet and the introduction of specific probiotics may assist with the management of specific types of chronic pain. This is an area that is currently undergoing more research, but has some exciting possibilities for the future of pain management.
How does the lining of our gut support our immune system?
Gut permeability is how easily nutrients, including proteins, move from your digestive system into the blood stream. This is a delicate balance where the intestinal wall plays guard, allowing properly digested microscopic food particles to move across into the blood stream, and be used to support our health and wellbeing. If the intestinal lining becomes highly permeable or “leaky”, then poorly digested, microscopic fragments of food may pass from the gut into the bloodstream causing issues with inflammation, food sensitivities, immune function and abdominal discomfort.

A healthy gut ensures our health and wellbeing, it also assists with the prevention of diseases of the gut and other organs within the body. There has been much research looking into how our gut and immune system are linked:
- Our microbiome is a teacher to our immune system: we are born with a very basic immune system and are protected by the antibodies we receive from our mothers. From here we develop our own antibodies from the food we eat, the environment we live and the bugs we fight off.
- Gut bacteria maintains immune system balance: our nose, lungs and gut constantly expose our immune system to “new things” such as pollen, dust, dirt and food. The role of our immune system is to identify what is “ok” and what needs to be fought off. The diverse range of bacteria, fungi and microorganisms that live within our gut, help the immune system to build a resilience to “new things”. The importance of the relationship is further highlighted by the fact 70-80% of our immune cells are found within our gut.
- The gut wall is a communication channel between our body and the microbiota: as discussed above the gut acts as a barrier permitting certain molecules to pass, whilst preventing others. The purpose of this is to support immune and brain function. The gut wall is made up of a:
- Physical barrier: epithelial cells that line the gut, the molecules on their surface and mucus they produce.
- Chemical barrier: inflammatory molecules (cytokines), antibodies, and antimicrobial substances produced by epithelial and immune cells.
- Preliminary findings on endotoxins and zonulin and inflammation:
- An endotoxin is a lipopolysaccharides (LPS) (large molecules found in some bacteria) which elicits a strong immune response when absorbed by the body. The endotoxin is released when the bacteria dies and cell wall breaks down. This can produce an immune response and the release of cytokines (which cause inflammation) when gut permeability allows movement of endotoxins into the blood supply.
- Zonulin is a protein produced by our body in response to gluten and specific bacteria, which has been linked to increasing gut permeability. When zonulin, endotoxins and cytokines enter the circulation they trigger an immune and inflammatory response.
What is the link between our gut, immune system and pain?
An interesting area of research has been looking at how gut permeability affects inflammation and pain, particularly with diseases such as irritable bowel syndrome (IBS). Scientists have noted that sensitisation of visceral afferent (sensory) pathways may contribute to pain and changes to the brain characterised by IBS. Some interesting findings include increased gut permeability, increased immune system activation, and an imbalance in descending pain inhibition. This is an interesting finding which may have applications for use in other inflammatory states, such as autoimmune diseases.
There has also been some preliminary studies looking at two painful conditions: fibromyalgia and complex regional pain syndrome and their relationship to gut permeability. These early findings have shown higher permeability in the gut of both conditions. Researchers are investigating how improving gut health may improve gut permeability and these painful conditions.
Almost 50% of the people living with chronic pain, also suffer from depression or anxiety. Researchers have been looking at the links between anxiety, depression and gut permeability. They have identified changes to the gut microbiome, including dysbiosis, as well as biomarkers for endotoxins and zonulin. Although this area of research requires more study, it has interesting applications in the area of mental health and pain.
A study released in 2020 “All disease begins in the (leaky) gut: role of zonulin-mediated gut permeability in the pathogenesis of some chronic inflammatory diseases” discusses the evolving science looking beyond genetic predisposition and environment, and towards the changes to gut permeability, immune activation, and changes to the microbiome, in chronic inflammatory diseases. The ongoing challenge in this area of science is determining if the changes occur as a result of the disease or are the cause of the disease, and whether improving the gut will improve the disease.
To continue reading Part 2 – The Gut-Brain Connection click here
Additional references
- Journal Article: Eubiosis and dysbiosis: the two sides of the microbiota (article here)
- Article: Our gut microbiome is always changing; it’s also remarkably stable (link here)
- Book: Gut Health and Probiotics. The science behind the hype. Jenny Tschiesche
- Book: Rushing Woman’s Syndrome. The impact of a never ending to-do list on your health. Dr Libby Weaver
- Book: Gut. The inside story of our body’s most under-rated organ. Giulia Enders
- Book: The mind-gut connection. How the hidden conversation within our bodies impacts on our mood, our choices, and our overall health. Emeran Mayer, MD
- Journal Article: The role of gut microbiota in immune homeostasis and autoimmunity (article here)
- Journal Article: The Role of Gut Microbiota in Intestinal Inflammation with Respect to Diet and Extrinsic Stressors (article here)
- Journal Article: Gut microbiota functions: metabolism of nutrients and other food components (article here)
- Journal Article: Stress and the gut microbiota-brain axis (article here)
- Journal Article: The gut microbiome: Relationships with disease and opportunities for therapy (article here)
- Journal Article: Neurotransmitter modulation by the gut microbiota (article here)
- Journal Article: Human nutrition, the gut microbiome and the immune system (article here)
- Journal Article: The microbiome: A key regulator of stress and neuroinflammation (article here)
- Journal Article: Current Understanding of Gut Microbiota in Mood Disorders: An Update of Human Studies (article here)
- Journal Article: Role of gut microbiota in brain function and stress-related pathology (article here)
- Journal Article: Intestinal permeability – a new target for disease prevention and therapy (article here)
- AUSMED E-Learning: Gut Microbiota Health and Wellbeing (link here)
- Journal Article: A review of lifestyle factors that contribute to important pathways associated with major depression: Diet, sleep and exercise (article here)
- Journal Article: ‘Gut health’: a new objective in medicine? (article here)
- Journal Article: Impact of the gut microbiota on inflammation, obesity, and metabolic disease (article here)